 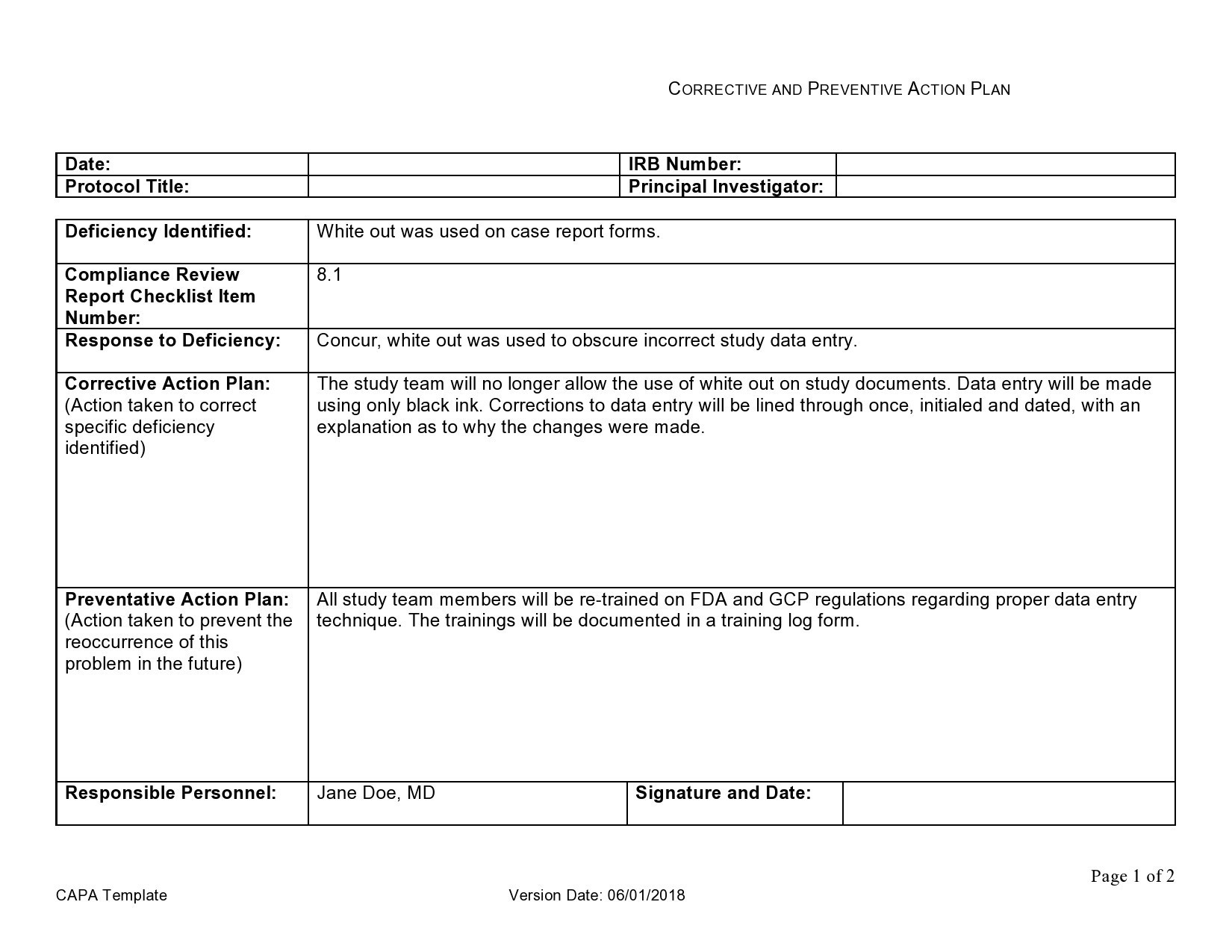 
Identify - Finally, the problem should be precisely identified and defined.Examine - A plan for identifying the problem must be worked out, preferably with a responsible person to identify the basic problem.These should be evaluated to identify the root cause of a problem. Analyze - The Capa process begins with a comprehensive analysis and documentation of all existing data.The following 7 steps are based on the defined FDA regulations regarding the CAPA report: The whole concept revolves around the idea of continuous improvement with the clear aim to reduce business expenditure and overhead.Īn effective CAPA report helps companies work efficiently and reduce the risk of errors and non-compliance with laws. It’s important to businesses because of its solution-oriented approach to standardizing operation procedures (SOP) so that tasks are made more efficient, employees are more productive, and failures are made more infrequent. 
From there the compliance officer will write up a corrective and preventative action plan. It is the job of the compliance officers to use a CAPA report form to comprehensively document a summary of the problem solution.Ī CAPA record contains information pertaining to a defect or deficiency found within the frame of operation, how the deficiency was dealt with, and who dealt with the problem. It includes an identification of the problem as well as the precise evaluation and implementation of solutions and corrective actions. to correct existing problems and prevent future errors from occurring. As you might have already guessed from its name - CAPA (Corrective and Preventative Action) - is a method that is used in a variety of settings such as businesses, healthcare, Economics, etc. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
